-
The table below shows properties of some elements A, B, C and D which belong to the same periodic table. The letters are not the actual symbols of the elements.
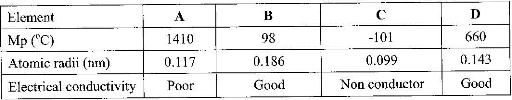 a) Arrange the elements in order they would appear in the periodic. Give a reason
b) Select the metallic element which is the better conductor of electricity. Give a reason.
a) Arrange the elements in order they would appear in the periodic. Give a reason
b) Select the metallic element which is the better conductor of electricity. Give a reason.
Date posted:
April 10, 2017
-
Four metal F, G, H and J were each separately added to cold water, hot water and steam. The table below is a summary of the observations made and the formulae of the hydroxides formed.
 a) Which two elements are likely to be in the same group of the periodic table?
b) Arrange the metals in the order of their reactivity starting with the most reactive.
a) Which two elements are likely to be in the same group of the periodic table?
b) Arrange the metals in the order of their reactivity starting with the most reactive.
Date posted:
April 10, 2017
-
The grid given below represents part of the periodic table. Study it and answer the questions that follow. (The letters do not represent the actual symbol of the elements)
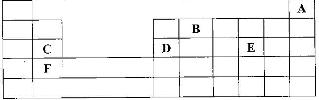 i) What name is given to the group of elements to which C and F belong?
ii) Which letter represents the element that is the least reactive?
iii) What type of bond is formed when B and E react? Explain
iv) Write the formula of the compound formed when element D and oxygen gas react
i) What name is given to the group of elements to which C and F belong?
ii) Which letter represents the element that is the least reactive?
iii) What type of bond is formed when B and E react? Explain
iv) Write the formula of the compound formed when element D and oxygen gas react
Date posted:
April 10, 2017
-
The table below gives the atomic numbers of elements W, X, Y and Z. The letters do not represent the actual symbols of the elements.
 a) Which one of the elements is least reactive? Explain.
b) (i) Which two elements would react most vigorously with each other?
(ii) Give the formula of the compound formed when the elements in b (i) react.
a) Which one of the elements is least reactive? Explain.
b) (i) Which two elements would react most vigorously with each other?
(ii) Give the formula of the compound formed when the elements in b (i) react.
Date posted:
April 7, 2017
-
The table below gives information on four elements represented by letters K, L, M and N. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.
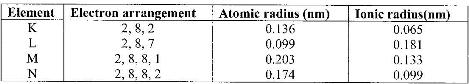 a) Which two elements have two similar properties? Explain.
b) What is the most likely formula of oxide of L?
c) Which element is a non-metal? Explain
d) Which one of the elements is the strongest reducing agent? Explain.
e) Explain why ionic radius of N is less than that of M.
f) Explain why the ionic radius of L is bigger than its atomic radius.
a) Which two elements have two similar properties? Explain.
b) What is the most likely formula of oxide of L?
c) Which element is a non-metal? Explain
d) Which one of the elements is the strongest reducing agent? Explain.
e) Explain why ionic radius of N is less than that of M.
f) Explain why the ionic radius of L is bigger than its atomic radius.
Date posted:
April 7, 2017
-
The table below gives the number of electrons, protons and neutrons in substances X, Y and Z. Study it and answer the questions that follow.
 a) Which letter represents an ion?
b) Which of the substances are isotopes? Give reason.
a) Which letter represents an ion?
b) Which of the substances are isotopes? Give reason.
Date posted:
April 7, 2017
-
The table below shows the number of valence electrons of the element P, Q and R.
 a) Explain why P and R would not be expected to form a compound.
b) Write an equation to show the effect of heat on the carbonate of R.
c) Write the formula for the most stable ion of Q
a) Explain why P and R would not be expected to form a compound.
b) Write an equation to show the effect of heat on the carbonate of R.
c) Write the formula for the most stable ion of Q
Date posted:
April 7, 2017
-
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.
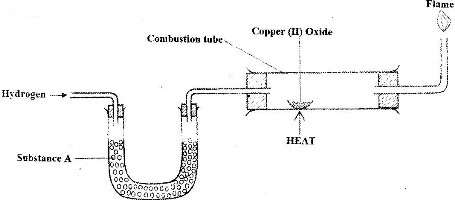 a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
Date posted:
April 7, 2017
-
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.
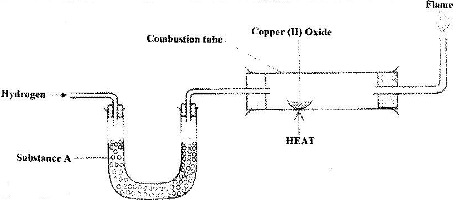 a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
a) Name substance A.
b) State the observation made in the combustion tube.
c) Explain the observation stated in (b) above.
Date posted:
April 7, 2017
-
The set-up below was used to investigate the products of burning biogas (methane). Study it and answer the questions that follow.
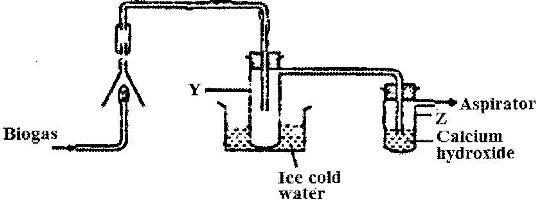 a) What product will be formed in test-tube Y?
b) State and explain the observations which will be made in Z.
a) What product will be formed in test-tube Y?
b) State and explain the observations which will be made in Z.
Date posted:
April 7, 2017
-
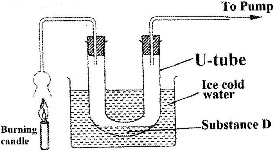
A student used the set up shown in the diagram below in order to study the reactions of some metals with steam. The experiment was carried out for ten minutes.
 a) What observation would be made if gas F is ignited?
b) When the experiment was repeated using iron powder instead of magnesium ribbon, very little gas F was obtained.
i) Give a reason for this observation.
ii) What change in the conditions of the experiment should the student have made in order to increase the volume of gas F produced?
a) What observation would be made if gas F is ignited?
b) When the experiment was repeated using iron powder instead of magnesium ribbon, very little gas F was obtained.
i) Give a reason for this observation.
ii) What change in the conditions of the experiment should the student have made in order to increase the volume of gas F produced?
Date posted:
April 7, 2017
-
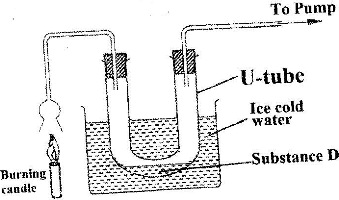
An experiment was set up as shown in the diagram below:
 a) Identify substance D.
b) Describe how the other product of the burning candle could be prevented from getting into the environment
a) Identify substance D.
b) Describe how the other product of the burning candle could be prevented from getting into the environment
Date posted:
April 7, 2017
-
In laboratory experiment hydrogen gas was passed over heated copper (II) oxide as shown in the diagram below.
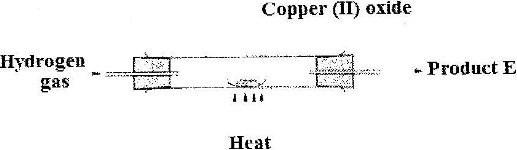 Describe a chemical test that can be used to identify the product E.
Describe a chemical test that can be used to identify the product E.
Date posted:
April 7, 2017
-
a) A candle wax is mainly a compound consisting of two elements. Name the two elements
b) The set-up below was used to investigate the burning of a candle. Study it and answer the questions that follow.
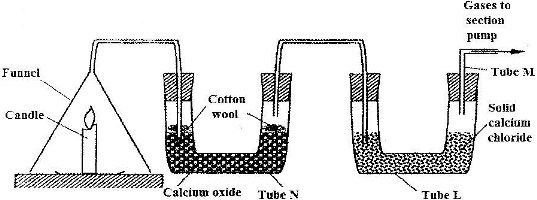 i) What would happen to the burning candle if the pump was turned off?
ii) State and explain the changes that are likely to occur in tube N by the end of the experiment.
iii) Name two gases that come out through tube M.
iv) What is the purpose of calcium chloride in tube L?
v) Name another substance that could be used in the place of calcium oxide in tube N.
i) What would happen to the burning candle if the pump was turned off?
ii) State and explain the changes that are likely to occur in tube N by the end of the experiment.
iii) Name two gases that come out through tube M.
iv) What is the purpose of calcium chloride in tube L?
v) Name another substance that could be used in the place of calcium oxide in tube N.
Date posted:
April 7, 2017
-
Study the set-up below and answer the questions that follow
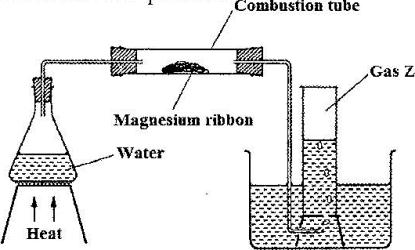 a) Write an equation for the reaction, which takes place in the combustion tube.
b) What property of gas Z allows it to be collected as shown in the diagram?
a) Write an equation for the reaction, which takes place in the combustion tube.
b) What property of gas Z allows it to be collected as shown in the diagram?
Date posted:
April 7, 2017
-
A student set up the experiment below to collect gas K. The gas K. The glass wool was heated before heating the zinc powder.
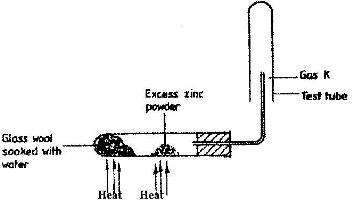 a) Why was it necessary to heat the moist glass wool before heating zinc powder?
b) What would happen if the zinc powder was heated before heating the glass wool?
c) What property of gas K makes it possible for it to be collected as shown in the diagram?
a) Why was it necessary to heat the moist glass wool before heating zinc powder?
b) What would happen if the zinc powder was heated before heating the glass wool?
c) What property of gas K makes it possible for it to be collected as shown in the diagram?
Date posted:
April 7, 2017
-
A measuring cylinder fitted with moist steel wool was inverted in a trough of water as shown in the diagram below.
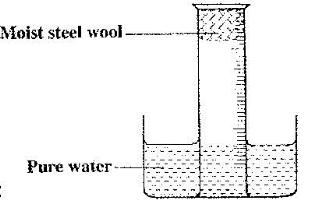 a) State and explain the observations made on the:
i) Moist steel wool after four days
ii) Water level in the measuring cylinder after four days.
b) What would be the effect of using steel wool moistened with salty water?
a) State and explain the observations made on the:
i) Moist steel wool after four days
ii) Water level in the measuring cylinder after four days.
b) What would be the effect of using steel wool moistened with salty water?
Date posted:
April 6, 2017
-
The set up below can be used to prepare oxygen gas. Study it and answer the questions that follow.
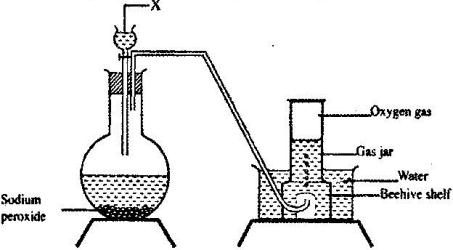 a) Identify X
b) What property of oxygen makes it possible for it to be collected as shown in the above set up?
c) State two uses of oxygen.
d) Write a balanced chemicals equation for the reaction that takes place in the reaction flask.
a) Identify X
b) What property of oxygen makes it possible for it to be collected as shown in the above set up?
c) State two uses of oxygen.
d) Write a balanced chemicals equation for the reaction that takes place in the reaction flask.
Date posted:
April 6, 2017
-
The following set up of three test-tubes was used to investigate rusting of iron. Study it and answer the questions that follow.
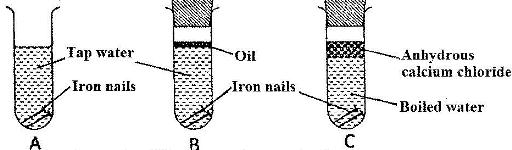
 a) Give a reason why rusting did not occur in test-tube C.
b) A luminium is used to protect iron sheets from rusting. Explain two ways in which aluminium protects iron from rusting.
a) Give a reason why rusting did not occur in test-tube C.
b) A luminium is used to protect iron sheets from rusting. Explain two ways in which aluminium protects iron from rusting.
Date posted:
April 6, 2017
-
In an experiment, dry hydrogen gas was passed over heated Lead (II) Oxide as shown in the diagram below.
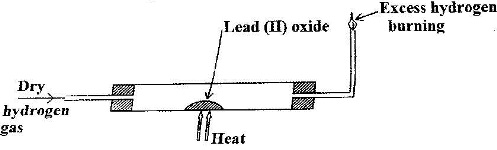 State and explain the observations made in the combustion tube
State and explain the observations made in the combustion tube
Date posted:
April 6, 2017
-
The diagram below represents a set-up that was used to show that part of air is used during burning.
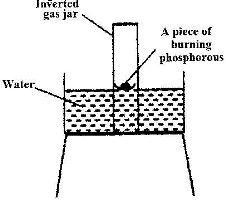 a) Given that phosphorus used was used in excess, draw a diagram of the set-up at the end of the experiment (when there was no further observable change).
b) Suggest one modification that should be made on the apparatus if the percentage of the air used is to be determined.
a) Given that phosphorus used was used in excess, draw a diagram of the set-up at the end of the experiment (when there was no further observable change).
b) Suggest one modification that should be made on the apparatus if the percentage of the air used is to be determined.
Date posted:
April 6, 2017
-
Air was passed through several reagents as shown in the flow chart below
 a) Write an equation for the reaction which takes place in the chamber with magnesium powder.
b) Name one gas which escapes from the chamber containing magnesium powder. Give reason for your answer.
a) Write an equation for the reaction which takes place in the chamber with magnesium powder.
b) Name one gas which escapes from the chamber containing magnesium powder. Give reason for your answer.
Date posted:
April 6, 2017
-
The diagram below is a set-up for the laboratory preparation of oxygen gas
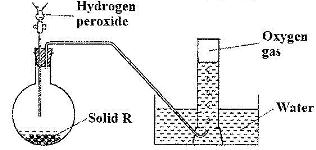 a) Name solid R.
b) Write an equation for the reaction that takes place in the flask.
c) Give one commercial use of oxygen.
a) Name solid R.
b) Write an equation for the reaction that takes place in the flask.
c) Give one commercial use of oxygen.
Date posted:
April 6, 2017
-
The diagram below shows an iron bar which supports a bridge. The iron bar is connected to a piece of magnesium metal.
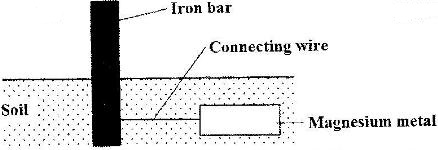 Explain why it is necessary to connect the piece of magnesium metal to the iron bar.
Explain why it is necessary to connect the piece of magnesium metal to the iron bar.
Date posted:
April 6, 2017
-
In an experiment a certain volume of air was passed repeatedly from syringe to syringe over heated excess zinc powder as shown in the diagram below.
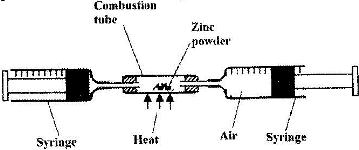 The experiment was repeated using excess magnesium powder. In which of two experiments was the change in the volume of air greatest? Give reasons.
The experiment was repeated using excess magnesium powder. In which of two experiments was the change in the volume of air greatest? Give reasons.
Date posted:
April 6, 2017
-
The set-Up below was used to study some properties of air.
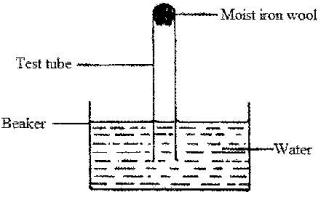 State and explain two observations that would be made at the end of the experiment.
State and explain two observations that would be made at the end of the experiment.
Date posted:
April 6, 2017
-
Study the diagram below and answer the questions that follow.
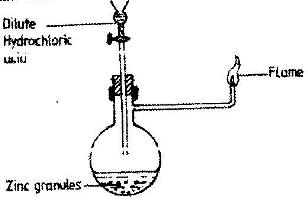 Write an equation for each of the two reactions that takes place in the experiment represented by the diagram
Write an equation for each of the two reactions that takes place in the experiment represented by the diagram
Date posted:
April 6, 2017
-
The graph below is a cooling curve for water. Study it and answer the questions that follow.
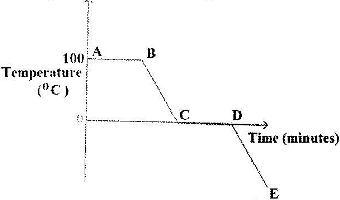 a) Explain what happens to the molecules of water in the region BC in terms of Kinetic theory.
b) In what state is the water in the region DE?
a) Explain what happens to the molecules of water in the region BC in terms of Kinetic theory.
b) In what state is the water in the region DE?
Date posted:
April 6, 2017
-
The set up below was used to separate a mixture of methanol and propanol. Study it and answer the questions that follow.
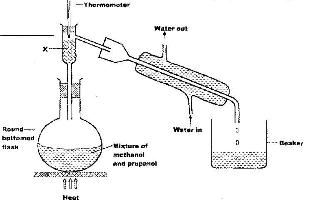 a) State the function of X.
b) Which liquid will collect first in the beaker? Given a reason.
a) State the function of X.
b) Which liquid will collect first in the beaker? Given a reason.
Date posted:
April 6, 2017
-
The chromatogram below was obtained from a contaminated food sample P. Contaminants Q,R,S and T are suspected to be in P. Use it to answer the following questions.
 a) Identify the contaminant in mixture P?
b) Which is the most soluble contaminant in P?
a) Identify the contaminant in mixture P?
b) Which is the most soluble contaminant in P?
Date posted:
April 6, 2017