-
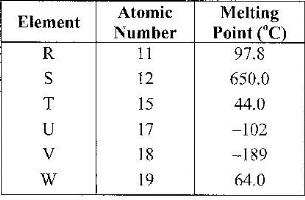
Use the information in the table below to answer the questions that follow. The letters do not represent the actual symbols of the elements
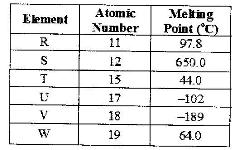 a) Give reasons why the melting point of:
(i) S is higher than that of R;
(ii) V is lower than of U.
b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain.
c) Write an equation for the reaction between T and excess oxygen.
d) Give one use element V.
a) Give reasons why the melting point of:
(i) S is higher than that of R;
(ii) V is lower than of U.
b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain.
c) Write an equation for the reaction between T and excess oxygen.
d) Give one use element V.
Date posted:
April 12, 2017
-
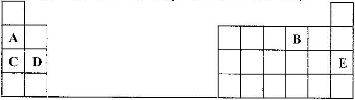
The grid given below represents part of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of elements
 (i) Select a letter which represents an element that looses electrons most readily. Given a reason for your answer
(ii) Explain why the atomic radius of P is found to be smaller than that of N.
(i) Select a letter which represents an element that looses electrons most readily. Given a reason for your answer
(ii) Explain why the atomic radius of P is found to be smaller than that of N.
Date posted:
April 12, 2017
-
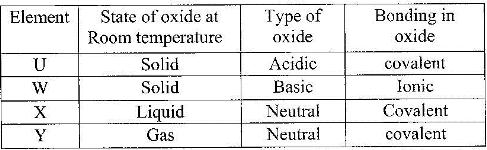
Use the information in the table below to answer the questions that follow. The letters do not represent the actual symbols of the elements.
 a) Give reasons why the melting point of:
i) S is higher than that of R;
ii) V is lower than that of U.
b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain
c) Write an equation for the reaction between T and excess oxygen.
d) Give one use of element V.
a) Give reasons why the melting point of:
i) S is higher than that of R;
ii) V is lower than that of U.
b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain
c) Write an equation for the reaction between T and excess oxygen.
d) Give one use of element V.
Date posted:
April 12, 2017
-
The chart below is part of the periodic table. Study it and answer the questions that follow. (The letters are not the actual symbols of the elements)
 i) Select the element in period three which has the shorter atomic radius. Give a reason for your answer
ii) Element F has the electronic structure, 2.8.18.4. On the chart above, indicate the position of element F.
iii) State one use of the elements of which E is a member.
iv) Write an equation to show the action of heat on the nitrate of element C.
i) Select the element in period three which has the shorter atomic radius. Give a reason for your answer
ii) Element F has the electronic structure, 2.8.18.4. On the chart above, indicate the position of element F.
iii) State one use of the elements of which E is a member.
iv) Write an equation to show the action of heat on the nitrate of element C.
Date posted:
April 12, 2017
-
The set-up below was used to prepare hydrogen gas
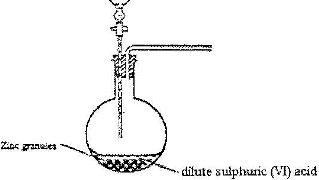 a) Write an equation for the reaction which takes place when hydrogen gas burns in air
b) State two industrial uses of hydrogen gas
a) Write an equation for the reaction which takes place when hydrogen gas burns in air
b) State two industrial uses of hydrogen gas
Date posted:
April 12, 2017
-
The set up shown below was used to investigate a property of hydrogen gas
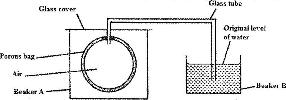 State and explain the observation that would be made in the glass tube if beaker A was filled with hydrogen gas.
State and explain the observation that would be made in the glass tube if beaker A was filled with hydrogen gas.
Date posted:
April 12, 2017
-
The graph below shows the relationship between pressure and temperature of a gas in a fixed volume container.
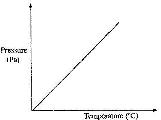 a) State the relationship between pressure and temperature that can be deduced from the graph.
b) Using kinetic theory, explain the relationship shown in the graph.
a) State the relationship between pressure and temperature that can be deduced from the graph.
b) Using kinetic theory, explain the relationship shown in the graph.
Date posted:
April 12, 2017
-
The graph below shows the behavior of a fixed mass of a gas at constant temperature.
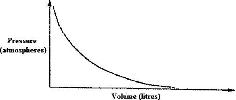 a) What is the relationship between the volume and the pressure of the gas?
b) 3 litres of oxygen gas at one atmosphere pressure were compressed to two atmospheres at constant temperature. Calculate the volume occupied by the oxygen gas
a) What is the relationship between the volume and the pressure of the gas?
b) 3 litres of oxygen gas at one atmosphere pressure were compressed to two atmospheres at constant temperature. Calculate the volume occupied by the oxygen gas
Date posted:
April 12, 2017
-
In an experiment to study the diffusion of gases, a student set up the apparatus shown in diagram 1. After sometimes the student noticed a change in the water level as shown in the diagram.
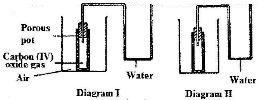 Give an explanation for the change in water level
Give an explanation for the change in water level
Date posted:
April 12, 2017
-
A mixture containing equal volumes of hydrogen and carbon (IV) oxide was introduced at one end of a tube as shown below.
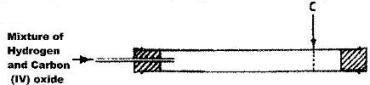 Which gas would be detected at point C first? Explain
Which gas would be detected at point C first? Explain
Date posted:
April 12, 2017
-
The diagram below shows a Jiko when in use. Study it and answer the questions that follow.
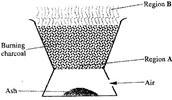 a) Identify the gas formed at region A
b) State and explain the observation made at region B.
a) Identify the gas formed at region A
b) State and explain the observation made at region B.
Date posted:
April 12, 2017
-
The diagram below represents part of a set-up used to prepare and collect gas T.
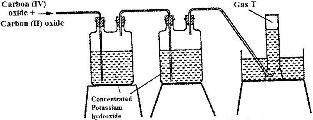 a) Name two reagents that are reacted to produce both carbon (IV) oxide and carbon (II) oxide
b) Write the equation for the reaction which takes place in the wash bottles
c) give a reason why carbon (II) oxide is not easily detected.
a) Name two reagents that are reacted to produce both carbon (IV) oxide and carbon (II) oxide
b) Write the equation for the reaction which takes place in the wash bottles
c) give a reason why carbon (II) oxide is not easily detected.
Date posted:
April 12, 2017
-
The set-up below was used to obtain a sample of iron
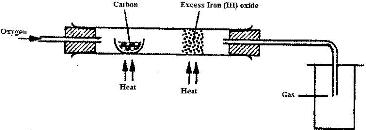 Write two equations for the reactions which occur in the combustion tube
Write two equations for the reactions which occur in the combustion tube
Date posted:
April 12, 2017
-
The set-up below was used to collect gas F, produced by the reaction between water and calcium metal
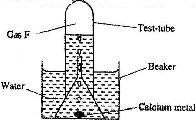 (i) Name gas F.
(ii) At the end of the experiment, the solution in the beaker was found to be a weak base. Explain why the solution is a weak base.
iii) Give one laboratory use of the solution formed in a beaker
(i) Name gas F.
(ii) At the end of the experiment, the solution in the beaker was found to be a weak base. Explain why the solution is a weak base.
iii) Give one laboratory use of the solution formed in a beaker
Date posted:
April 12, 2017
-
Carbon (II) oxide was passed over heated iron (III) oxide as shown in the diagram below.
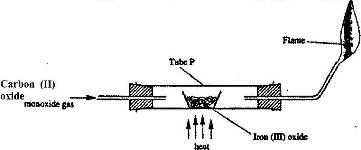 a) Give the observation made in tube P.
b) Write the equation for the reaction which takes place in tube P.
a) Give the observation made in tube P.
b) Write the equation for the reaction which takes place in tube P.
Date posted:
April 12, 2017
-
When steam was passed over heated charcoal as shown in the diagram below, hydrogen and carbon (II) oxide gases were formed.
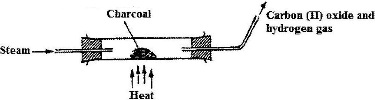 a) Write the equation for the reaction which takes place.
b) Name two uses of carbon (II) oxide gas, which are also uses of hydrogen gas.
a) Write the equation for the reaction which takes place.
b) Name two uses of carbon (II) oxide gas, which are also uses of hydrogen gas.
Date posted:
April 12, 2017
-
The apparatus shown below was used to investigate the effect of carbon (II) oxide on copper (II) oxide.
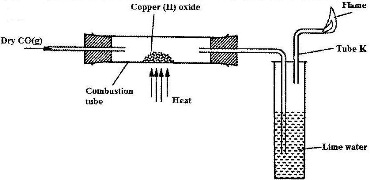 a) State the observation that was made in the combustion tube at the end of the experiment
b) Write an equation for the reaction that took place in the combustion tube.
c) why is it necessary to burn the gas coming out of tube K.
a) State the observation that was made in the combustion tube at the end of the experiment
b) Write an equation for the reaction that took place in the combustion tube.
c) why is it necessary to burn the gas coming out of tube K.
Date posted:
April 12, 2017
-
In an experiment, carbon (IV) oxide gas was passed over heated charcoal and the gas produced collected as shown in the diagram below.
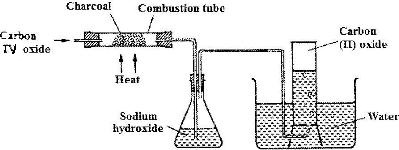 i) Write an equation for the reaction that took place in the combustion tube.
ii) Name another substance that can be used instead of sodium hydroxide
iii) Describe a simple chemical test that can be used to distinguish between carbon (IV) oxide and carbon (II) oxide.
iv) Give one use of carbon (II) oxide.
i) Write an equation for the reaction that took place in the combustion tube.
ii) Name another substance that can be used instead of sodium hydroxide
iii) Describe a simple chemical test that can be used to distinguish between carbon (IV) oxide and carbon (II) oxide.
iv) Give one use of carbon (II) oxide.
Date posted:
April 10, 2017
-
The following diagrams show the structures of two allotropes of carbon. Study them and answer the questions that follow.
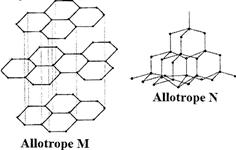 Allotrope M
(i) Name allotrope M and N
(ii) Give one use of N
(iii) Which allotrope conduct electricity? Explain.
Allotrope M
(i) Name allotrope M and N
(ii) Give one use of N
(iii) Which allotrope conduct electricity? Explain.
Date posted:
April 10, 2017
-
The simplified flow chart shows some of the steps in the manufacture of sodium carbonate by the Solvay process.
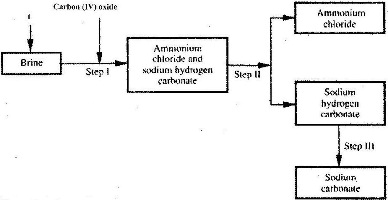 a) Identify substance L.
b) Name the process taking place in step II.
c) Write an equation for the reaction, which takes place in step III.
a) Identify substance L.
b) Name the process taking place in step II.
c) Write an equation for the reaction, which takes place in step III.
Date posted:
April 10, 2017
-
The diagram below represents a charcoal burner. Study it and answer the question that follows:
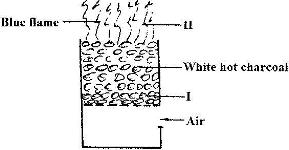
Date posted:
April 10, 2017
-
A student investigated the effect of an electric current by passing it through some substances. The student used inert electrodes, and connected a bulb to the circuit. The table below shows the substances used and their states.
 a) In which experiment did the bulb not light?
b) Explain your answer in (a) above.
a) In which experiment did the bulb not light?
b) Explain your answer in (a) above.
Date posted:
April 10, 2017
-
The table below gives information about elements A1, A2, A3 and A4
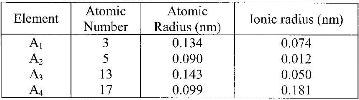 (i) In which period of the periodic table is element A2? Give a reason.
ii) Explain why the atomic radius of:
I. A1 is greater than that of A2;
II. A4 is smaller than its ionic radius.
iii) Select the element which is in the same group as A3.
(i) In which period of the periodic table is element A2? Give a reason.
ii) Explain why the atomic radius of:
I. A1 is greater than that of A2;
II. A4 is smaller than its ionic radius.
iii) Select the element which is in the same group as A3.
Date posted:
April 10, 2017
-
The diagram below illustrates a method of preparing salts by direct synthesis.
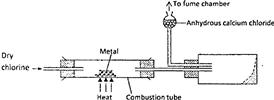 a) This method can be used to prepare either aluminium chloride or iron (III) chloride. Explain why it cannot be used to prepare sodium chloride.
b) Describe how a sample of sodium can be prepared in the laboratory by direct synthesis.
a) This method can be used to prepare either aluminium chloride or iron (III) chloride. Explain why it cannot be used to prepare sodium chloride.
b) Describe how a sample of sodium can be prepared in the laboratory by direct synthesis.
Date posted:
April 10, 2017
-
The set-up below was used to prepare anhydrous chlorides of elements in laboratory where no fume cupboard was available. The chlorides were to be collected in flask 1.
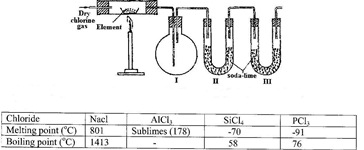 The following table shows the melting and boiling points of the chlorides that were prepared.
a) Explain why it is necessary to pass dry chlorine through the apparatus before heating each element.
b) Give two reasons why tubes II and III were filled with soda lime (solid mixture of sodium hydroxide and calcium hydroxide)
c) Explain why it would not be possible to collect any sodium chloride in flask 1
d) Name one other substance that can be used in tubes II and III
e) Write an equation for the reaction that forms phosphorous (III) chloride
f) Describe how you would separate a mixture of sodium chloride and aluminium chloride
The following table shows the melting and boiling points of the chlorides that were prepared.
a) Explain why it is necessary to pass dry chlorine through the apparatus before heating each element.
b) Give two reasons why tubes II and III were filled with soda lime (solid mixture of sodium hydroxide and calcium hydroxide)
c) Explain why it would not be possible to collect any sodium chloride in flask 1
d) Name one other substance that can be used in tubes II and III
e) Write an equation for the reaction that forms phosphorous (III) chloride
f) Describe how you would separate a mixture of sodium chloride and aluminium chloride
Date posted:
April 10, 2017
-
a) Other than their location in the atom, name two other differences between an electron and a proton.
b) The table below gives the number of electrons, protons and neutrons in particles
A, B, C, D, E, F and G.
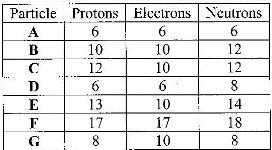 i) Which particle is likely to be a halogen?
ii) What is the mass number of E?
iii) Write a formula of the compound formed when E combines with G.
iv) Name the type of bond formed in (iii) above.
v) How does the radii of C and E compare? Give a reason.
vi) Draw a dot (.) and cross (x) diagram for the compound formed between A and F
vii) Why would particle B not react with particle D?
i) Which particle is likely to be a halogen?
ii) What is the mass number of E?
iii) Write a formula of the compound formed when E combines with G.
iv) Name the type of bond formed in (iii) above.
v) How does the radii of C and E compare? Give a reason.
vi) Draw a dot (.) and cross (x) diagram for the compound formed between A and F
vii) Why would particle B not react with particle D?
Date posted:
April 10, 2017
-
The diagram below shows the bonding between aluminium chloride and ammonia.
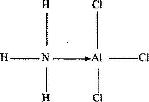 a) Name the types of bonds that exists in the molecule
b) How many electrons are used for bonding in the molecule?
a) Name the types of bonds that exists in the molecule
b) How many electrons are used for bonding in the molecule?
Date posted:
April 10, 2017
-
The table below gives atomic numbers of elements represented by the letters A, B, C and D.
 Use the information to answer the questions that follow.
a) Name the type of bonding that exists in the compound formed when A and D react.
b) Select the letter which represents the best oxidizing agent. Give a reason for your answer.
Use the information to answer the questions that follow.
a) Name the type of bonding that exists in the compound formed when A and D react.
b) Select the letter which represents the best oxidizing agent. Give a reason for your answer.
Date posted:
April 10, 2017
-
The diagram below is a section of a model of the structure of element T.
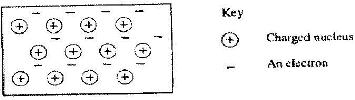 a) State the type of bonding that exists in T.
b) In which group of the periodic table does element T belong? Give a reason.
a) State the type of bonding that exists in T.
b) In which group of the periodic table does element T belong? Give a reason.
Date posted:
April 10, 2017
-
Study the information given in the table below and answer the questions that follow.
The letters do not represent the actual symbols of the elements
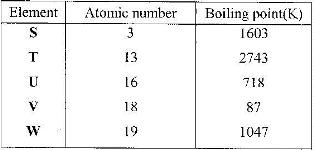 a) Select the elements which belong to the same:
i) Group
ii) Period
b) Which element
i) is in gaseous state at room temperature? Explain. (Take room temperature to be 298K)
ii) Does not form an oxide?
c) Write the
i) Formula of the nitrate of element T.
ii) Equation for the reaction between elements S and U
d) What type of bond would exists in the compound formed when U and T react? Give a reason for your answer
e) The aqueous sulphate of element W was electrolyzed using inert electrodes. Name products formed at the:
i) Cathode
ii) Anode
a) Select the elements which belong to the same:
i) Group
ii) Period
b) Which element
i) is in gaseous state at room temperature? Explain. (Take room temperature to be 298K)
ii) Does not form an oxide?
c) Write the
i) Formula of the nitrate of element T.
ii) Equation for the reaction between elements S and U
d) What type of bond would exists in the compound formed when U and T react? Give a reason for your answer
e) The aqueous sulphate of element W was electrolyzed using inert electrodes. Name products formed at the:
i) Cathode
ii) Anode
Date posted:
April 10, 2017